The study found that the Coronavirus can remain infectious on surfaces for the duration of DAYS by transforming into a microscopic pie-like film.
- More than 99.9% of the fluid in coronavirus droplets evaporates within minutes
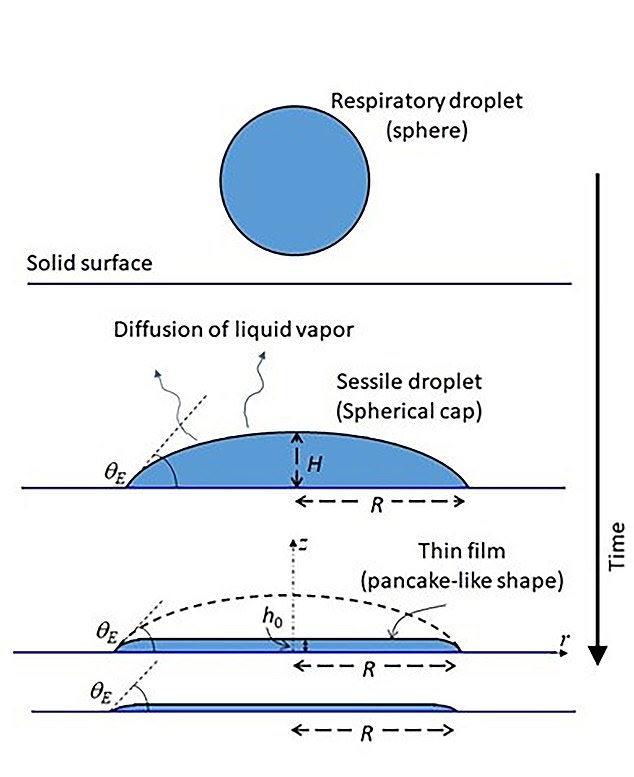
- But the virus survives because the dried droplet turns into a pie-like film
- This sticks to the surface for hours and can still be contagious if touched
- Scientists recommend cleaning surfaces regularly to ensure there is no covid
Coronavirus-containing particles that land on a surface remain infectious for several days by transforming into a microscopic pie-like film after the water in the droplet evaporates.
This conversion sees more than 99.9 percent of the droplet liquid disappearing within minutes, but the virus lives in a protective layer of the remaining liquid.
Small forces keep the film, which is only nanometers thick, clings to the surface and slows the evaporation process.
The film completely evaporates at different times, depending on the material it landed on, with a large stainless steel and copper drop lasting only 24 and 16 hours, respectively.
But it can live for more than 150 hours on polypropylene. The smallest drop, about a tenth of the size, lasts more than 80 hours on glass.
These numbers are based on experiments conducted in laboratory conditions, and are likely to be lower in the real world where there are variable amounts of heat and air flow – factors that promote evaporation.
Scroll down for the video
The film (lower part) covers the same area as the droplet (top), with the same radius and initial gradient angle. The only measurement that changes is its vertical height
Rajneesh Bhardwaj and Amit Agrawal, professors at IIT Bombay, specialize in using computer models and physics to understand how coronavirus droplets can spread disease.
And they previously found that wearing a face mask reduces the size of the withdrawal of infectious corona virus particles resulting from coughing by up to 23 times.

The particles containing the Coronavirus from sneezing and coughing that land on an infectious surface remain for several hours because the water in the droplet evaporates and turns from a ball into a microscopic membrane that looks like a pancake (stock)
In movie form, the coronavirus can endure for hours, even days, on hydrophobic surfaces if left uninterrupted.
The film covers the same area as the drop, with the same radius and initial gradient angle. The only measurement that changes is its vertical height.
Data from the study published in Fluid Physics, Found that the thickness of the film slowly decreases even and throughout this time the amount of coronavirus in the droplet / film is constant.
“The biggest surprise to us was that the drying time of this nanoscale film was within hours,” said Bhardwaj.
It indicates that the surface is not completely dry, and that the slowly evaporating nanometer layer provides the medium required for the survival of the coronavirus.
The researchers say their findings indicate that the virus can survive in a viable form for an extended period of time, and they emphasized the need for regular and thorough cleaning of surfaces.
“It is advisable to disinfect frequently touched surfaces, such as doorknobs or handheld devices, and inside hospitals and other areas prone to outbreaks,” Agrawal said.
We also recommend heating the surfaces, because even short-term high temperatures, in which the surface is at a higher temperature than the ambient temperature, can help evaporate the nanometer layer and destroy the virus.

Communicator. Reader. Hipster-friendly introvert. General zombie specialist. Tv trailblazer